Your cart is currently empty!

27334 – 2 CM 2 Vectra Sound Heads are now $195.00 WHILE SUPPLIES LAST
We Have All Chattanooga brand Parts, please call if you do not see the part you need!
Please send us an email at: office@chattmed.com or use our RFQ tool “Request for Quote button”
with any inquiries into prices for items not displayed.
Your Chattanooga Group Brand Products Experts!
FREE SHIPPING OVER $1500.00*
*Tables, Some Hydrocollators and other items marked as “Freight” are not applicable for free shipping.
Chattanooga Medical Supply offers Regular and Specialty Line units.
Specialty Chattanooga Machine Line Units Include but are not limited to:
- Vectra Genisys
- laser units
- Vectra Sound Heads
- GALAXY 300 & 400 TableS
- Diodes for lasers 820 1040NM
- Diathermy units
- Carts for int. leg 2 units.
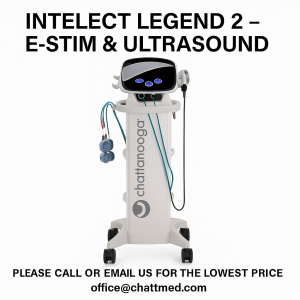
- Intelect Legend 2 transport & intelect legend 2 combo units.
- Repairs
*NEW* Intelect legend 2 and transport 2 available!
We have the LOWEST prices on 2″ round, 2″ square and 2″ x 3.5″ electrodes*